| Reference | 1. J Mol Model. 2013 Jan;19(1):119-30. doi: 10.1007/s00894-012-1528-7. Epub 2012 Jul 22.<br />
Structure-based design of nitrogen-linked macrocyclic kinase inhibitors leading to the clinical candidate SB1317/TG02, a potent inhibitor of cyclin dependant kinases (CDKs), Janus kinase 2 (JAK2), and Fms-like tyrosine kinase-3 (FLT3).<br />
Poulsen A(1), William A, Blanchard S, Nagaraj H, Williams M, Wang H, Lee A, Sun E, Teo EL, Tan E, Goh KC, Dymock B.<br />
Author information:<br />
(1)S*BIO Pte Ltd, 1 Science Park Road, #05-09 The Capricorn, Singapore Science Park II, Singapore, 117 528, Singapore. [email protected]<br />
A high-throughput screen against Aurora A kinase revealed several promising submicromolar pyrimidine-aniline leads. The bioactive conformation found by docking these leads into the Aurora A ATP-binding site had a semicircular shape. Macrocycle formation was proposed to achieve novelty and selectivity via ring-closing metathesis of a diene precursor. The nature of the optimal linker and its size was directed by docking. In a kinase panel screen, selected macrocycles were active on other kinase targets, mainly FLT3, JAK2, and CDKs. These compounds then became leads in a CDK/FLT3/JAK2 inhibitor project. Macrocycles with a basic nitrogen in the linker form a salt bridge with Asp86 in CDK2 and Asp698 in FLT3. Interaction with this residue explains the observed selectivity. The Asp86 residue is conserved in most CDKs, resulting in potent pan-CDK inhibition by these compounds. Optimized macrocycles generally have good DMPK properties, and are efficacious in mouse models of cancer. Compound 5 (SB1317/TG02), a pan-CDK/FLT3/JAK2 inhibitor, was selected for preclinical development, and is now in phase 1 clinical trials.<br />
2. Drug Metab Lett. 2012 Mar;6(1):33-42.<br />
Preclinical metabolism and pharmacokinetics of SB1317 (TG02), a potent CDK/JAK2/FLT3 inhibitor.<br />
Pasha MK(1), Jayaraman R, Reddy VP, Yeo P, Goh E, Williams A, Goh KC, Kantharaj E.<br />
Author information:<br />
(1)S*BIO Pte Ltd, 1 Science Park Road, No. 05-09, The Capricorn, Singapore Science Park II, Singapore.<br />
SB1317 (TG02) is a novel small molecule potent CDK/JAK2/FLT3 inhibitor. To evaluate full potential of this development candidate, we conducted drug metabolism and pharmacokinetic studies of this novel anti-cancer agent. SB1317 was soluble, highly permeable in Caco-2 cells, and showed > 99% binding to plasma from mice, dog and humans. It was metabolically stable in human and dog liver microsomes relative to mouse and rat. SB1317 was mainly metabolized by CYP3A4 and CY1A2 in vitro. SB1317 did not inhibit any of the major human CYPs in vitro except CYP2D6 (IC50=1 μM). SB1317 did not significantly induce CYP1A and CYP3A4 in human hepatocytes in vitro. The metabolic profiles in liver microsomes from preclinical species were qualitatively similar to humans. In pharmacokinetic studies SB1317 showed moderate to high systemic clearance (relative to liver blood flow), high volume of distribution ( > 0.6 L/kg), oral bioavailability of 24%, ~ 4 and 37% in mice, rats and dogs, respectively; and extensive tissue distribution in mice. The favorable ADME of SB1317 supported its preclinical development as an oral drug candidate.<br />
3. J Med Chem. 2012 Jan 12;55(1):169-96. doi: 10.1021/jm201112g. Epub 2011 Dec 29.<br />
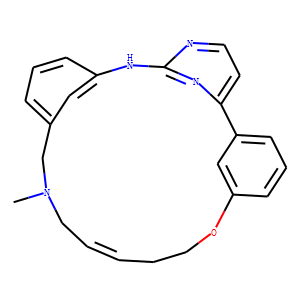
Discovery of kinase spectrum selective macrocycle (16E)-14-methyl-20-oxa-5,7,14,26-tetraazatetracyclo[19.3.1.1(2,6).1(8,12)]heptaco sa-1(25),2(26),3,5,8(27),9,11,16,21,23-decaene (SB1317/TG02), a potent inhibitor of cyclin dependent kinases (CDKs), Janus kinase 2 (JAK2), and fms-like tyrosine kinase-3 (FLT3) for the treatment of cancer.<br />
William AD(1), Lee AC, Goh KC, Blanchard S, Poulsen A, Teo EL, Nagaraj H, Lee CP, Wang H, Williams M, Sun ET, Hu C, Jayaraman R, Pasha MK, Ethirajulu K, Wood JM, Dymock BW.<br />
Author information:<br />
(1)S BIO Pte. Ltd., 1 Science Park Road, No. 05-09, The Capricorn, Singapore Science Park II, 117528, Singapore. [email protected]<br />
Herein, we describe the design, synthesis, and SAR of a series of unique small molecule macrocycles that show spectrum selective kinase inhibition of CDKs, JAK2, and FLT3. The most promising leads were assessed in vitro for their inhibition of cancer cell proliferation, solubility, CYP450 inhibition, and microsomal stability. This screening cascade revealed 26 h as a preferred compound with target IC(50) of 13, 73, and 56 nM for CDK2, JAK2 and FLT3, respectively. Pharmacokinetic (PK) studies of 26 h in preclinical species showed good oral exposures. Oral efficacy was observed in colon (HCT-116) and lymphoma (Ramos) xenograft studies, in line with the observed PK/PD correlation. 26h (SB1317/TG02) was progressed into development in 2010 and is currently undergoing phase 1 clinical trials in advanced leukemias and multiple myeloma.<br />
|