| InChI | InChI=1S/C22H29ClN4O6/c1-22(2,3)18(26-19(31)12-6-7-15(24)14(23)9-12)21(33)27-8-4-5-16(27)20(32)25-13(11-28)10-17(29)30/h6-7,9,11,13,16,18H,4-5,8,10,24H2,1-3H3,(H,25,32)(H,26,31)(H,29,30)/t13-,16-,18+/m0/s1 |
| Reference | 1. J Pharmacol Exp Ther. 2007 May;321(2):509-16. Epub 2007 Feb 8.
<br>
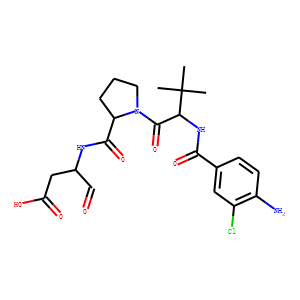
(S)-1-((S)-2-{[1-(4-amino-3-chloro-phenyl)-methanoyl]-amino}-3,3-dimethyl-butanoy
l)-pyrrolidine-2-carboxylic acid
((2R,3S)-2-ethoxy-5-oxo-tetrahydro-furan-3-yl)-amide (VX-765), an orally
available selective interleukin (IL)-converting enzyme/caspase-1 inhibitor,
exhibits potent anti-inflammatory activities by inhibiting the release of
IL-1beta and IL-18.
<br>
Wannamaker W(1), Davies R, Namchuk M, Pollard J, Ford P, Ku G, Decker C,
Charifson P, Weber P, Germann UA, Kuida K, Randle JC.
<br>
Author information: <br>
(1)Department of Chemistry, Drug Discovery Support Unit, Vertex Pharmaceuticals,
Inc., 130 Waverly St., Cambridge, MA 02139, USA.
<br>
(S)-1-((S)-2-{[1-(4-amino-3-chloro-phenyl)-methanoyl]-amino}-3,3-dimethyl-butanoy
l)-pyrrolidine-2-carboxylic acid
((2R,3S)-2-ethoxy-5-oxo-tetrahydro-furan-3-yl)-amide (VX-765) is an orally
absorbed prodrug of
(S)-3-({1-[(S)-1-((S)-2-{[1-(4-amino-3-chlorophenyl)-methanoyl]-amino}-3,3-dimeth
yl-butanoyl)-pyrrolidin-2yl]-methanoyl}-amino)-4-oxo-butyric acid (VRT-043198), a
potent and selective inhibitor of interleukin-converting enzyme/caspase-1
subfamily caspases. VRT-043198 exhibits 100- to 10,000-fold selectivity against
other caspase-3 and -6 to -9. The therapeutic potential of VX-765 was assessed by
determining the effects of VRT-043198 on cytokine release by monocytes in vitro
and of orally administered VX-765 in several animal models in vivo. In cultures
of peripheral blood mononuclear cells and whole blood from healthy subjects
stimulated with bacterial products, VRT-043198 inhibited the release of
interleukin (IL)-1beta and IL-18, but it had little effect on the release of
several other cytokines, including IL-1alpha, tumor necrosis factor-alpha, IL-6
and IL-8. In contrast, VRT-043198 had little or no demonstrable activity in
cellular models of apoptosis, and it did not affect the proliferation of
activated primary T cells or T-cell lines. VX-765 was efficiently converted to
VRT-043198 when administered orally to mice, and it inhibited
lipopolysaccharide-induced cytokine secretion. In addition, VX-765 reduced
disease severity and the expression of inflammatory mediators in models of
rheumatoid arthritis and skin inflammation. These data suggest that VX-765 is a
novel cytokine inhibitor useful for treatment of inflammatory diseases.
<br>
|