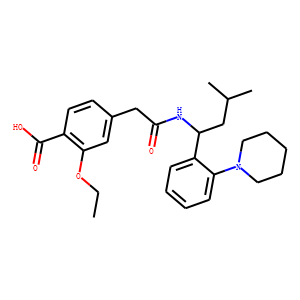
| InChI | InChI=1S/C27H36N2O4/c1-4-33-25-17-20(12-13-22(25)27(31)32)18-26(30)28-23(16-19(2)3)21-10-6-7-11-24(21)29-14-8-5-9-15-29/h6-7,10-13,17,19,23H,4-5,8-9,14-16,18H2,1-3H3,(H,28,30)(H,31,32)/t23-/m0/s1 |
| Reference | 1: Tanaka K, Okada Y, Mori H, Torimoto K, Arao T, Tanaka Y. The Effects of Mitiglinide and Repaglinide on Postprandial Hyperglycemia in Patients Undergoing Methylprednisolone Pulse Therapy. Intern Med. 2017 Oct 11. doi: 10.2169/internalmedicine.9013-17. [Epub ahead of print] PubMed PMID: 29021480.<br />
2: Shinde UA, Modani SH, Singh KH. Design and Development of Repaglinide Microemulsion Gel for Transdermal Delivery. AAPS PharmSciTech. 2017 Jul 17. doi: 10.1208/s12249-017-0811-4. [Epub ahead of print] PubMed PMID: 28717973.<br />
3: Wei H, Zhou T, Tan B, Zhang L, Li M, Xiao Z, Xu F. Impact of chronic unpredicted mild stress-induced depression on repaglinide fate via glucocorticoid signaling pathway. Oncotarget. 2017 Jul 4;8(27):44351-44365. doi: 10.18632/oncotarget.17874. PubMed PMID: 28574832; PubMed Central PMCID: PMC5546485.<br />
4: Kim SJ, Toshimoto K, Yao Y, Yoshikado T, Sugiyama Y. Quantitative Analysis of Complex Drug-Drug Interactions Between Repaglinide and Cyclosporin A/Gemfibrozil Using Physiologically Based Pharmacokinetic Models With In Vitro Transporter/Enzyme Inhibition Data. J Pharm Sci. 2017 Sep;106(9):2715-2726. doi: 10.1016/j.xphs.2017.04.063. Epub 2017 May 4. PubMed PMID: 28479356.<br />
5: Xiao ZX, Chen RQ, Hu DX, Xie XQ, Yu SB, Chen XQ. Identification of repaglinide as a therapeutic drug for glioblastoma multiforme. Biochem Biophys Res Commun. 2017 Jun 17;488(1):33-39. doi: 10.1016/j.bbrc.2017.04.157. Epub 2017 May 2. PubMed PMID: 28476618.<br />
6: Kalehoei E, Azadbakht M. The beneficial effect of repaglinide on in vitro maturation and development ability of immature mouse oocytes. In Vitro Cell Dev Biol Anim. 2017 Aug;53(7):626-631. doi: 10.1007/s11626-017-0152-3. Epub 2017 Apr 21. PubMed PMID: 28432599.<br />
7: Kancherla P, Keesari S, Alegete P, Khagga M, Das P. Identification, isolation, and synthesis of seven novel impurities of anti-diabetic drug Repaglinide. Drug Test Anal. 2017 Apr 19. doi: 10.1002/dta.2207. [Epub ahead of print] PubMed PMID: 28421713.<br />
8: Kudo T, Goda H, Yokosuka Y, Tanaka R, Komatsu S, Ito K. Estimation of the Contribution of CYP2C8 and CYP3A4 in Repaglinide Metabolism by Human Liver Microsomes Under Various Buffer Conditions. J Pharm Sci. 2017 Sep;106(9):2847-2852. doi: 10.1016/j.xphs.2017.02.013. Epub 2017 Feb 24. PubMed PMID: 28238899.<br />
9: Awasthi R, Kulkarni GT, Ramana MV, de Jesus Andreoli Pinto T, Kikuchi IS, Molim Ghisleni DD, de Souza Braga M, De Bank P, Dua K. Dual crosslinked pectin-alginate network as sustained release hydrophilic matrix for repaglinide. Int J Biol Macromol. 2017 Apr;97:721-732. doi: 10.1016/j.ijbiomac.2017.01.050. Epub 2017 Jan 20. PubMed PMID: 28115226.<br />
10: Amin MM, Arbid MS. Estimation of ellagic acid and/or repaglinide effects on insulin signaling, oxidative stress, and inflammatory mediators of liver, pancreas, adipose tissue, and brain in insulin resistant/type 2 diabetic rats. Appl Physiol Nutr Metab. 2017 Feb;42(2):181-192. doi: 10.1139/apnm-2016-0429. Epub 2016 Oct 21. PubMed PMID: 28092161.
|