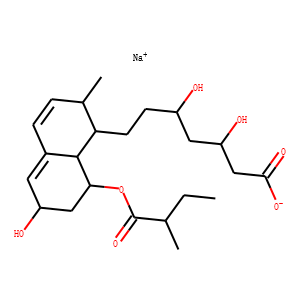
| InChI | InChI=1S/C23H36O7.Na/c1-4-13(2)23(29)30-20-11-17(25)9-15-6-5-14(3)19(22(15)20)8-7-16(24)10-18(26)12-21(27)28;/h5-6,9,13-14,16-20,22,24-26H,4,7-8,10-12H2,1-3H3,(H,27,28);/q;+1/p-1/t13-,14-,16+,17+,18+,19-,20-,22-;/m0./s1 |
| Reference | </br>1:Design of fixed dose combination and physicochemical characterization of enteric-coated bilayer tablet with circadian rhythmic variations containing telmisartan and pravastatin sodium. Luo D, Kim JH, Park C, Oh E, Park JB, Cui JH, Cao QR, Lee BJ.Int J Pharm. 2017 May 15;523(1):343-356. doi: 10.1016/j.ijpharm.2017.03.030. Epub 2017 Mar 19. PMID: 28330645 </br>2:Formulation and Pharmacokinetic Evaluation of Microcapsules Containing Pravastatin Sodium Using Rats. Puttegowda VD, Karki R, Goli D, Jha SK, Mudagal MP.Scientifica (Cairo). 2016;2016:7623193. doi: 10.1155/2016/7623193. Epub 2016 Aug 9. PMID: 27595040 Free PMC Article</br>3:Duodenum-triggered delivery of pravastatin sodium: II. Design, appraisal and pharmacokinetic assessments of enteric surface-decorated nanocubosomal dispersions. Tayel SA, El-Nabarawi MA, Tadros MI, Abd-Elsalam WH.Drug Deliv. 2016 Nov;23(9):3266-3278. Epub 2016 Apr 19. PMID: 27094305 </br>4:A laboratory data-based evaluation of the efficacy and safety of generic pravastatin sodium for long-term use. Suzuki M, Kanamori M, Hashimoto T, Hashimoto Y, Funakoshi R, Sasaki T.J Pharm Health Care Sci. 2015 Dec 18;2:1. doi: 10.1186/s40780-015-0033-4. eCollection 2016. PMID: 26819746 Free PMC Article</br>5:[Preparation and performance of pravastatin sodium-loaded chitosan microspheres]. Feng J, Liu Z, Yan Y, Hu M, Lu L, You W.Nan Fang Yi Ke Da Xue Xue Bao. 2015 Jun;35(6):879-82. Chinese. PMID: 26111689 </br>6:Duodenum-triggered delivery of pravastatin sodium via enteric surface-coated nanovesicular spanlastic dispersions: development, characterization and pharmacokinetic assessments. Tayel SA, El-Nabarawi MA, Tadros MI, Abd-Elsalam WH.Int J Pharm. 2015 Apr 10;483(1-2):77-88. doi: 10.1016/j.ijpharm.2015.02.012. Epub 2015 Feb 7. PMID: 25666025 </br>7:Pravastatin sodium. Al-Badr AA, Mostafa GA.Profiles Drug Subst Excip Relat Methodol. 2014;39:433-513. doi: 10.1016/B978-0-12-800173-8.00008-8. Review. PMID: 24794911 </br>8:[Simultaneous determination of repaglinide and pravastatin sodium in rat plasma by LC-ms/MS and its application on pharmacokinetic interactions study]. Ma YR, Zhou Y, Zhang GQ, Rao Z, Huang J, Wei YH, Wu XA.Yao Xue Xue Bao. 2014 Jan;49(1):72-7. Chinese. PMID: 24783509 </br>9:Biopolymeric mucoadhesive bilayer patch of pravastatin sodium for buccal delivery and treatment of patients with atherosclerosis. Yedurkar P, Dhiman MK, Petkar K, Sawant K.Drug Dev Ind Pharm. 2013 May;39(5):670-80. doi: 10.3109/03639045.2012.687379. Epub 2012 May 26. PMID: 22630116 </br>10:Bilayered transmucosal drug delivery system of pravastatin sodium: statistical optimization, in vitro, ex vivo, in vivo and stability assessment. Maurya SK, Bali V, Pathak K.Drug Deliv. 2012 Jan;19(1):45-57. doi: 10.3109/10717544.2011.644348. Epub 2011 Dec 23. PMID: 22191789
|