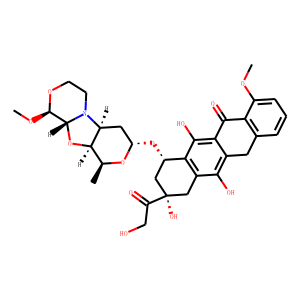
| IUPAC Name | (7S,9S)-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-7-[[(2S,4R,6S,7S,9R,10S)-10-methoxy-6-methyl-5,8,11-trioxa-1-azatricyclo[7.4.0.02,7]tridecan-4-yl]oxy]-8,10-dihydro-7H-tetracene-5,12-dione |
| InChI | InChI=1S/C32H35NO13/c1-13-29-16(33-7-8-43-31(42-3)30(33)46-29)9-20(44-13)45-18-11-32(40,19(35)12-34)10-15-22(18)28(39)24-23(26(15)37)25(36)14-5-4-6-17(41-2)21(14)27(24)38/h4-6,13,16,18,20,29-31,34,37,39-40H,7-12H2,1-3H3/t13-,16-,18-,20-,29+,30+,31-,32-/m0/s1 |
| Reference | </br>1:Virtual Cross-Linking of the Active Nemorubicin Metabolite PNU-159682 to Double-Stranded DNA. Scalabrin M, Quintieri L, Palumbo M, Riccardi Sirtori F, Gatto B.Chem Res Toxicol. 2017 Feb 20;30(2):614-624. doi: 10.1021/acs.chemrestox.6b00362. Epub 2017 Jan 25. PMID: 28068470 </br>2:The interaction of nemorubicin metabolite PNU-159682 with DNA fragments d(CGTACG)(2), d(CGATCG)(2) and d(CGCGCG)(2) shows a strong but reversible binding to G:C base pairs. Mazzini S, Scaglioni L, Mondelli R, Caruso M, Sirtori FR.Bioorg Med Chem. 2012 Dec 15;20(24):6979-88. doi: 10.1016/j.bmc.2012.10.033. Epub 2012 Nov 3. PMID: 23154079 </br>3:In vitro hepatic conversion of the anticancer agent nemorubicin to its active metabolite PNU-159682 in mice, rats and dogs: a comparison with human liver microsomes. Quintieri L, Fantin M, Palatini P, De Martin S, Rosato A, Caruso M, Geroni C, Floreani M.Biochem Pharmacol. 2008 Sep 15;76(6):784-95. doi: 10.1016/j.bcp.2008.07.003. Epub 2008 Jul 11. PMID: 18671948 </br>4:Formation and antitumor activity of PNU-159682, a major metabolite of nemorubicin in human liver microsomes. Quintieri L, Geroni C, Fantin M, Battaglia R, Rosato A, Speed W, Zanovello P, Floreani M.Clin Cancer Res. 2005 Feb 15;11(4):1608-17. PMID: 15746066 Free Article
|