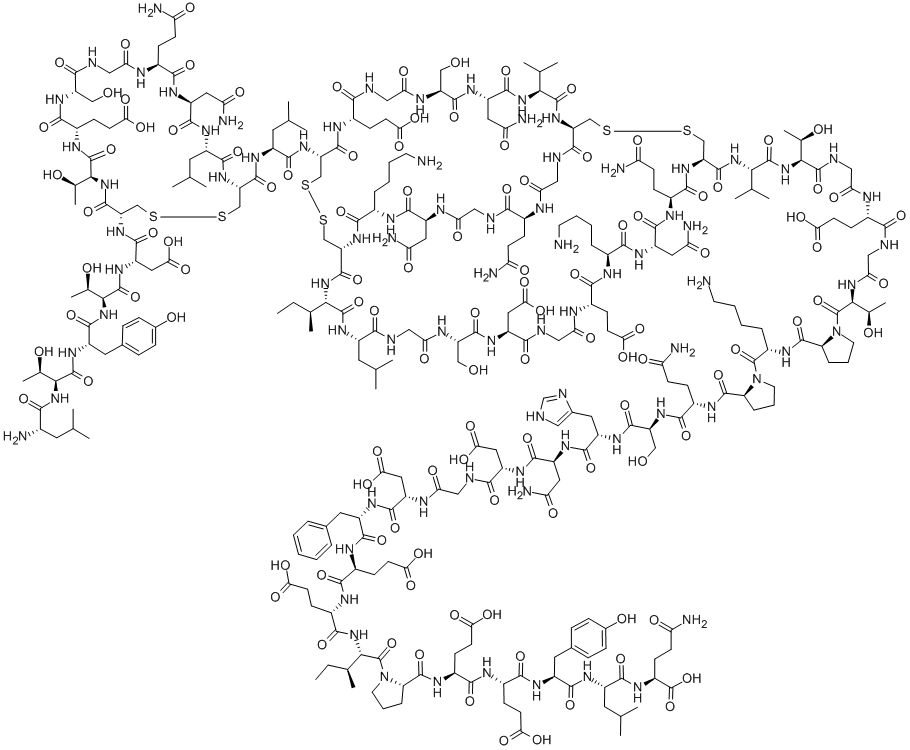
| IUPAC Name | (2S)-5-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-1-[(2S,3S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[2-[[(2S)-2-[[(2S)-4-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-amino-2-[[(2S)-1-[(2S)-6-amino-2-[[(2S)-1-[(2S,3R)-2-[[2-[[(2S)-2-[[2-[[(2S,3R)-2-[[(2S)-2-[[(1R,6R,9S,12S,15S,18S,24S,27S,33S,36S,39R,44R,47S,53S,56S,59S,67S,73S,76S)-15,76-bis(4-aminobutyl)-44-[[(2S)-2-[[(4R,7S,10S,13S,19S,22S,25S,28R)-28-[[(2S)-2-[[(2S,3R)-2-[[(2S)-2-[[(2S,3R)-2-[[(2S)-2-amino-4-methylpentanoyl]amino]-3-hydroxybutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-3-hydroxybutanoyl]amino]-3-carboxypropanoyl]amino]-10-(2-amino-2-oxoethyl)-13-(3-amino-3-oxopropyl)-22-(2-carboxyethyl)-25-[(1R)-1-hydroxyethyl]-19-(hydroxymethyl)-7-(2-methylpropyl)-6,9,12,15,18,21,24,27-octaoxo-1,2-dithia-5,8,11,14,17,20,23,26-octazacyclononacosane-4-carbonyl]amino]-4-methylpentanoyl]amino]-12,56,73-tris(2-amino-2-oxoethyl)-9,67-bis(3-amino-3-oxopropyl)-36-[(2S)-butan-2-yl]-18,47-bis(2-carboxyethyl)-24-(carboxymethyl)-27,53-bis(hydroxymethyl)-33-(2-methylpropyl)-8,11,14,17,20,23,26,29,32,35,38,45,48,51,54,57,60,62,65,68,71,74,77-tricosaoxo-59-propan-2-yl-3,4,41,42-tetrathia-7,10,13,16,19,22,25,28,31,34,37,46,49,52,55,58,61,63,66,69,72,75,78-tricosazabicyclo[37.22.17]octaheptacontane-6-carbonyl]amino]-3-methylbutanoyl]amino]-3-hydroxybutanoyl]amino]acetyl]amino]-4-carboxybutanoyl]amino]acetyl]amino]-3-hydroxybutanoyl]pyrrolidine-2-carbonyl]amino]hexanoyl]pyrrolidine-2-carbonyl]amino]-5-oxopentanoyl]amino]-3-hydroxypropanoyl]amino]-3-(1H-imidazol-4-yl)propanoyl]amino]-4-oxobutanoyl]amino]-3-carboxypropanoyl]amino]acetyl]amino]-3-carboxypropanoyl]amino]-3-phenylpropanoyl]amino]-4-carboxybutanoyl]amino]-4-carboxybutanoyl]amino]-3-methylpentanoyl]pyrrolidine-2-carbonyl]amino]-4-carboxybutanoyl]amino]-4-carboxybutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-methylpentanoyl]amino]-5-oxopentanoic acid |
| InChI | InChI=1S/C287H440N80O111S6/c1-24-132(17)225-280(470)345-162(87-126(5)6)236(426)306-111-209(396)320-181(116-370)267(457)344-176(101-220(416)417)238(428)307-105-203(390)316-152(61-74-213(402)403)243(433)321-146(40-29-32-80-288)241(431)339-171(96-198(298)385)259(449)326-153(56-69-194(294)381)250(440)351-186(121-482-479-118-183-240(430)310-107-202(389)313-148(54-67-192(292)379)233(423)303-108-206(393)317-170(95-197(297)384)258(448)322-147(41-30-33-81-289)242(432)350-187(272(462)359-225)122-483-480-119-184(269(459)323-150(60-73-212(400)401)235(425)304-110-208(395)319-180(115-369)266(456)342-174(99-201(301)388)265(455)357-223(130(13)14)278(468)355-183)352-254(444)165(90-129(11)12)335-270(460)185-120-481-484-123-188(354-263(453)178(103-222(420)421)347-283(473)230(137(22)375)362-264(454)168(93-141-48-52-144(378)53-49-141)346-282(472)228(135(20)373)361-232(422)145(291)86-125(3)4)273(463)363-229(136(21)374)281(471)330-158(65-78-217(410)411)249(439)348-179(114-368)239(429)309-106-204(391)315-151(55-68-193(293)380)244(434)340-172(97-199(299)386)260(450)334-164(89-128(9)10)253(443)353-185)271(461)358-224(131(15)16)279(469)364-227(134(19)372)277(467)311-112-205(392)314-149(59-72-211(398)399)234(424)305-113-210(397)356-231(138(23)376)286(476)367-85-37-45-191(367)276(466)331-160(42-31-34-82-290)284(474)365-83-35-43-189(365)274(464)328-154(57-70-195(295)382)248(438)349-182(117-371)268(458)338-169(94-142-104-302-124-312-142)257(447)341-173(98-200(300)387)261(451)343-175(100-219(414)415)237(427)308-109-207(394)318-177(102-221(418)419)262(452)337-166(91-139-38-27-26-28-39-139)255(445)327-155(62-75-214(404)405)245(435)325-159(66-79-218(412)413)251(441)360-226(133(18)25-2)285(475)366-84-36-44-190(366)275(465)329-157(64-77-216(408)409)246(436)324-156(63-76-215(406)407)247(437)336-167(92-140-46-50-143(377)51-47-140)256(446)333-163(88-127(7)8)252(442)332-161(287(477)478)58-71-196(296)383/h26-28,38-39,46-53,104,124-138,145-191,223-231,368-378H,24-25,29-37,40-45,54-103,105-123,288-291H2,1-23H3,(H2,292,379)(H2,293,380)(H2,294,381)(H2,295,382)(H2,296,383)(H2,297,384)(H2,298,385)(H2,299,386)(H2,300,387)(H2,301,388)(H,302,312)(H,303,423)(H,304,425)(H,305,424)(H,306,426)(H,307,428)(H,308,427)(H,309,429)(H,310,430)(H,311,467)(H,313,389)(H,314,392)(H,315,391)(H,316,390)(H,317,393)(H,318,394)(H,319,395)(H,320,396)(H,321,433)(H,322,448)(H,323,459)(H,324,436)(H,325,435)(H,326,449)(H,327,445)(H,328,464)(H,329,465)(H,330,471)(H,331,466)(H,332,442)(H,333,446)(H,334,450)(H,335,460)(H,336,437)(H,337,452)(H,338,458)(H,339,431)(H,340,434)(H,341,447)(H,342,456)(H,343,451)(H,344,457)(H,345,470)(H,346,472)(H,347,473)(H,348,439)(H,349,438)(H,350,432)(H,351,440)(H,352,444)(H,353,443)(H,354,453)(H,355,468)(H,356,397)(H,357,455)(H,358,461)(H,359,462)(H,360,441)(H,361,422)(H,362,454)(H,363,463)(H,364,469)(H,398,399)(H,400,401)(H,402,403)(H,404,405)(H,406,407)(H,408,409)(H,410,411)(H,412,413)(H,414,415)(H,416,417)(H,418,419)(H,420,421)(H,477,478)/t132-,133-,134+,135+,136+,137+,138+,145-,146-,147-,148-,149-,150-,151-,152-,153-,154-,155-,156-,157-,158-,159-,160-,161-,162-,163-,164-,165-,166-,167-,168-,169-,170-,171-,172-,173-,174-,175-,176-,177-,178-,179-,180-,181-,182-,183-,184-,185-,186-,187-,188-,189-,190-,191-,223-,224-,225-,226-,227-,228-,229-,230-,231-/m0/s1 |
| Reference | 1.<span style="font-family: Arial, sans-serif; font-size: 13px; font-variant-ligatures: normal; orphans: 2; widows: 2;">Greinacher, Andreas, and Norbert Lubenow. "Recombinant hirudin in clinical practice: focus on lepirudin." </span><i style="font-family: Arial, sans-serif; font-size: 13px; font-variant-ligatures: normal; orphans: 2; widows: 2;">Circulation</i><span style="font-family: Arial, sans-serif; font-size: 13px; font-variant-ligatures: normal; orphans: 2; widows: 2;"> 103.10 (2001): 1479-1484.<br />
2.</span><span style="font-family: Arial, sans-serif; font-size: 13px; font-variant-ligatures: normal; orphans: 2; widows: 2;">Greinacher, Andreas, et al. "Lepirudin (recombinant hirudin) for parenteral anticoagulation in patients with heparin-induced thrombocytopenia." </span><i style="font-family: Arial, sans-serif; font-size: 13px; font-variant-ligatures: normal; orphans: 2; widows: 2;">Circulation</i><span style="font-family: Arial, sans-serif; font-size: 13px; font-variant-ligatures: normal; orphans: 2; widows: 2;"> 100.6 (1999): 587-593.<br />
3.</span>Expert Rev Cardiovasc Ther. 2004 May;2(3):339-57. doi: 10.1586/14779072.2.3.339. Lepirudin: a bivalent direct thrombin inhibitor for anticoagulation therapy.Greinacher A(1).
<div>
</div>
<div>
Lepirudin (Refludan), Berlex Laboratories, USA and Canada; Pharmion, all other countries), a recombinant derivative of the naturally occurring leech anticoagulant hirudin, was the first direct thrombin inhibitor to be approved by the European Agency for the Evaluation of Medicinal Products and the US Food and Drug Administration for the treatment of heparin-induced thrombocytopenia. Since its introduction into Europe and the USA, it has been studied in over 7000 patients requiring anticoagulation in conditions including acute coronary syndromes, percutaneous coronary intervention, cardiopulmonary bypass and heparin-induced thrombocytopenia. Three European clinical trials, designated eparin-Associated Thrombocytopenia (HAT)-1, -2 and -3, demonstrated the efficacy and safety of lepirudin in the prevention and treatment of thrombosis in patients with antibody-confirmed heparin-induced thrombocytopenia. A postmarketing, observational study, termed the Drug-Monitoring Program, evaluated lepirudin in over 1000 patients with heparin-induced thrombocytopenia in the setting of routine clinical practice. In the Drug-Monitoring Program, adverse events were substantially reduced compared with clinical trials, while clinical efficacy was maintained; suggesting that insight gained through clinical experience was translated into improved safety. Here, pharmacotherapy using lepirudin is reviewed, with particular reference to clinical studies in heparin-induced thrombocytopenia, and some recommendations based on this extensive clinical experience with lepirudin are provided. Although only approved for the treatment of heparin-induced thrombocytopenia, the use of lepirudin in acute coronary syndromes, percutaneous coronary intervention, vascular surgery and coronary artery bypass grafting is also discussed. The review concludes with a discussion of pharmacokinetic and clinical data supporting the potential for subcutaneous administration of lepirudin.</div>
<div>
</div>
|