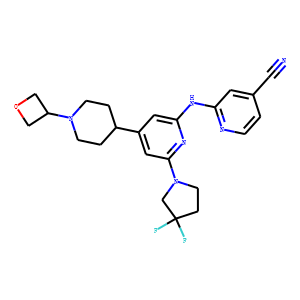
| InChI | InChI=1S/C23H26F2N6O/c24-23(25)4-8-31(15-23)22-11-18(17-2-6-30(7-3-17)19-13-32-14-19)10-21(29-22)28-20-9-16(12-26)1-5-27-20/h1,5,9-11,17,19H,2-4,6-8,13-15H2,(H,27,28,29) |
| Reference | 1. J Med Chem. 2015 Jan 8;58(1):401-18. doi: 10.1021/jm5013984. Epub 2014 Oct 23. <br />
Discovery of dual leucine zipper kinase (DLK, MAP3K12) inhibitors with activity
in neurodegeneration models. <br />
Patel S(1), Cohen F, Dean BJ, De La Torre K, Deshmukh G, Estrada AA, Ghosh AS,
Gibbons P, Gustafson A, Huestis MP, Le Pichon CE, Lin H, Liu W, Liu X, Liu Y, Ly
CQ, Lyssikatos JP, Ma C, Scearce-Levie K, Shin YG, Solanoy H, Stark KL, Wang J,
Wang B, Zhao X, Lewcock JW, Siu M. <br />
Author information: <br />
(1)Departments of †Discovery Chemistry, ‡Neurosciences, §Drug Metabolism and
Pharmacokinetics, and ∥Biochemical and Cellular Pharmacology, Genentech, Inc. , 1
DNA Way, South San Francisco, California 94080, United States. <br />
Dual leucine zipper kinase (DLK, MAP3K12) was recently identified as an essential
regulator of neuronal degeneration in multiple contexts. Here we describe the
generation of potent and selective DLK inhibitors starting from a high-throughput
screening hit. Using proposed hinge-binding interactions to infer a binding mode
and specific design parameters to optimize for CNS druglike molecules, we came to
focus on the di(pyridin-2-yl)amines because of their combination of desirable
potency and good brain penetration following oral dosing. Our lead inhibitor
GNE-3511 (26) displayed concentration-dependent protection of neurons from
degeneration in vitro and demonstrated dose-dependent activity in two different
animal models of disease. These results suggest that specific pharmacological
inhibition of DLK may have therapeutic potential in multiple indications. <br />
DOI: 10.1021/jm5013984 <br />
PMID: 25341110 [Indexed for MEDLINE]
|