| Reference | 1. J Med Chem. 2017 May 11;60(9):3580-3590. doi: 10.1021/acs.jmedchem.7b00032. Epub
2017 May 1. <br />
<br />
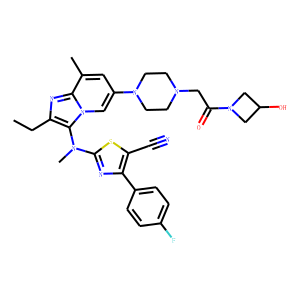
Discovery of
2-[[2-Ethyl-6-[4-[2-(3-hydroxyazetidin-1-yl)-2-oxoethyl]piperazin-1-yl]-8-methyli
midazo[1,2-a]pyridin-3-yl]methylamino]-4-(4-fluorophenyl)thiazole-5-carbonitrile
(GLPG1690), a First-in-Class Autotaxin Inhibitor Undergoing Clinical Evaluation
for the Treatment of Idiopathic Pulmonary Fibrosis. <br />
<br />
Desroy N(1), Housseman C(1), Bock X(1), Joncour A(1), Bienvenu N(1), Cherel L(1),
Labeguere V(1), Rondet E(1), Peixoto C(1), Grassot JM(1), Picolet O(1), Annoot
D(1), Triballeau N(1), Monjardet A(1), Wakselman E(1), Roncoroni V(1), Le Tallec
S(1), Blanque R(1), Cottereaux C(1), Vandervoort N(2), Christophe T(2), Mollat
P(1), Lamers M(3), Auberval M(1), Hrvacic B(4), Ralic J(4), Oste L(2), van der
Aar E(2), Brys R(2), Heckmann B(1). <br />
<br />
Author information: <br />
(1)Galapagos SASU , 102 Avenue Gaston Roussel, 93230 Romainville, France.
(2)Galapagos NV , Generaal De Wittelaan L11 A3, 2800 Mechelen, Belgium.
(3)Charles River Laboratories , Chesterford Research Park, CB10 1XL Saffron
Walden, United Kingdom.
(4)Fidelta Ltd. , Prilaz baruna Filipovića 29, Zagreb, HR-10000, Croatia. <br />
<br />
Autotaxin is a circulating enzyme with a major role in the production of
lysophosphatic acid (LPA) species in blood. A role for the autotaxin/LPA axis has
been suggested in many disease areas including pulmonary fibrosis. Structural
modifications of the known autotaxin inhibitor lead compound 1, to attenuate hERG
inhibition, remove CYP3A4 time-dependent inhibition, and improve pharmacokinetic
properties, led to the identification of clinical candidate GLPG1690 (11).
Compound 11 was able to cause a sustained reduction of LPA levels in plasma in
vivo and was shown to be efficacious in a bleomycin-induced pulmonary fibrosis
model in mice and in reducing extracellular matrix deposition in the lung while
also reducing LPA 18:2 content in bronchoalveolar lavage fluid. Compound 11 is
currently being evaluated in an exploratory phase 2a study in idiopathic
pulmonary fibrosis patients.
|