| Catalog Number | I004382 |
| CAS Number | 63223-86-9 |
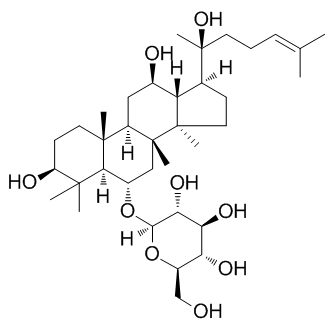
| Molecular Formula | C36H62O9
|
| Purity | 95% |
| Solubility | 10 mM in DMSO |
| Storage | Store at -20°C |
| IUPAC Name | (2R,3R,4S,5S,6R)-2-[[(3S,5R,6S,8R,9R,10R,12R,13R,14R,17S)-3,12-dihydroxy-17-[(2S)-2-hydroxy-6-methylhept-5-en-2-yl]-4,4,8,10,14-pentamethyl-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-6-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol |
| InChI | InChI=1S/C36H62O9/c1-19(2)10-9-13-36(8,43)20-11-15-34(6)26(20)21(38)16-24-33(5)14-12-25(39)32(3,4)30(33)22(17-35(24,34)7)44-31-29(42)28(41)27(40)23(18-37)45-31/h10,20-31,37-43H,9,11-18H2,1-8H3/t20-,21+,22-,23+,24+,25-,26-,27+,28-,29+,30-,31+,33+,34+,35+,36-/m0/s1 |
| InChIKey | RAQNTCRNSXYLAH-RFCGZQMISA-N |
| SMILES | CC(=CCC[C@@](C)([C@H]1CC[C@@]2([C@@H]1[C@@H](C[C@H]3[C@]2(C[C@@H]([C@@H]4[C@@]3(CC[C@@H](C4(C)C)O)C)O[C@H]5[C@@H]([C@H]([C@@H]([C@H](O5)CO)O)O)O)C)O)C)O)C |
| Reference | <p style=/line-height:25px/>
<br>[1]. Lee SY, et al. Anti-inflammatory effects of ginsenoside Rg1 and its metabolites ginsenoside Rh1 and 20(S)-protopanaxatriol in mice with TNBS-induced colitis. Eur J Pharmacol. 2015 Jun 6;762:333-343.
<br>[2]. Feng Y, et al. Ginsenoside Rh1 Improves the Effect of Dexamethasone on Autoantibodies Production and Lymphoproliferation in MRL/lpr Mice. Evid Based Complement Alternat Med. 2015;2015:727650.
<br>[3]. Siddiqi MH, et al. Ginsenoside Rh1 induces mouse osteoblast growth and differentiation through the bone morphogenetic protein 2/runt-related gene 2 signalling pathway. J Pharm Pharmacol. 2014 Dec;66(12):1763-73.
<br>[4]. Fayeza M. Siraj, et al. In silico screening of ginsenoside Rh1 with PPARγ and in vitro analysis on 3T3-L1 cell line. Molecular Simulation
Volume 41, Issue 15, 2015
</p>
|