| Catalog Number | I000420 |
| CAS Number | 11021-13-9 |
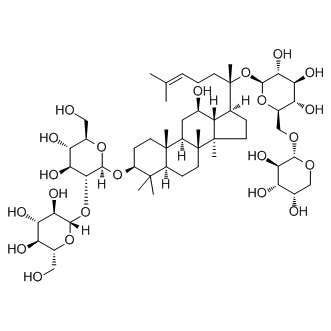
| Molecular Formula | C53H90O22
|
| Purity | 95% |
| Solubility | 10 mM in water |
| Storage | Store at -20°C |
| IUPAC Name | (2S,3R,4S,5S,6R)-2-[(2R,3R,4S,5S,6R)-4,5-dihydroxy-6-(hydroxymethyl)-2-[[(3S,5R,8R,9R,10R,12R,13R,14R,17S)-12-hydroxy-4,4,8,10,14-pentamethyl-17-[(2S)-6-methyl-2-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2S,3R,4S,5S)-3,4,5-trihydroxyoxan-2-yl]oxymethyl]oxan-2-yl]oxyhept-5-en-2-yl]-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-yl]oxy]oxan-3-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol |
| InChI | InChI=1S/C53H90O22/c1-23(2)10-9-14-53(8,75-47-43(67)39(63)37(61)29(72-47)22-69-45-41(65)34(58)26(57)21-68-45)24-11-16-52(7)33(24)25(56)18-31-50(5)15-13-32(49(3,4)30(50)12-17-51(31,52)6)73-48-44(40(64)36(60)28(20-55)71-48)74-46-42(66)38(62)35(59)27(19-54)70-46/h10,24-48,54-67H,9,11-22H2,1-8H3/t24-,25+,26-,27+,28+,29+,30-,31+,32-,33-,34-,35+,36+,37+,38-,39-,40-,41+,42+,43+,44+,45-,46-,47-,48-,50-,51+,52+,53-/m0/s1 |
| InChIKey | NODILNFGTFIURN-GZPRDHCNSA-N |
| SMILES | CC(=CCCC(C)(C1CCC2(C1C(CC3C2(CCC4C3(CCC(C4(C)C)OC5C(C(C(C(O5)CO)O)O)OC6C(C(C(C(O6)CO)O)O)O)C)C)O)C)OC7C(C(C(C(O7)COC8C(C(C(CO8)O)O)O)O)O)O)C |
| Reference | <p style=/line-height:25px/>
<br>[1]. Huang Q, et al. Ginsenoside-Rb2 displays anti-osteoporosis effects through reducing oxidative damage and bone-resorbing cytokines during osteogenesis. Bone. 2014 Sep;66:306-14.
<br>[2]. Yoo YC, et al. Protective effect of ginsenoside-Rb2 from Korean red ginseng on the lethal infection of haemagglutinating virus of Japan in mice. J Ginseng Res. 2013 Mar;37(1):80-6.
<br>[3]. Lim HJ, et al. Inhibitory effects of ginsenoside-rb2 on nicotinic stimulation-evoked catecholamine secretion. Korean J Physiol Pharmacol. 2014 Oct;18(5):431-9.
</p>
|