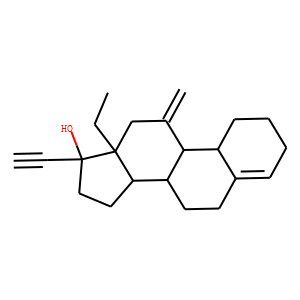
| IUPAC Name | (8S,9S,10R,13S,14S,17R)-13-ethyl-17-ethynyl-11-methylidene-1,2,3,6,7,8,9,10,12,14,15,16-dodecahydrocyclopenta[a]phenanthren-17-ol |
| Reference | 1: Park MJ, Jeon GH. Pulmonary embolism in a healthy woman using the oral contraceptives containing desogestrel. Obstet Gynecol Sci. 2017 Mar;60(2):232-235. doi: 10.5468/ogs.2017.60.2.232. Epub 2017 Mar 16. PubMed PMID: 28344968; PubMed Central PMCID: PMC5364109.<br />
2: Tanmahasamut P, Saejong R, Rattanachaiyanont M, Angsuwathana S, Techatraisak K, Sanga-Areekul N. Postoperative desogestrel for pelvic endometriosis-related pain: a randomized controlled trial. Gynecol Endocrinol. 2017 Jul;33(7):534-539. doi: 10.1080/09513590.2017.1296124. Epub 2017 Mar 7. PubMed PMID: 28266234.<br />
3: Merki-Feld GS, Imthurn B, Dubey R, Sándor PS, Gantenbein AR. Improvement of migraine with change from combined hormonal contraceptives to progestin-only contraception with desogestrel: How strong is the effect of taking women off combined contraceptives? J Obstet Gynaecol. 2017 Apr;37(3):338-341. doi: 10.1080/01443615.2016.1234438. Epub 2017 Jan 27. PubMed PMID: 28129717.<br />
4: Peters K, Gordon N, Ricciotti N, Hsieh J, Howard B, Weiss H. Hemostatic effects of two desogestrel-containing combined oral contraceptive regimens: a multinational, multicenter, randomized, open-label study. Clin Exp Obstet Gynecol. 2016;43(3):334-40. PubMed PMID: 27328486.<br />
5: Joubert F, Perrin-Terrin AS, Verkaeren E, Cardot P, Fiamma MN, Frugière A, Rivals I, Similowski T, Straus C, Bodineau L. Desogestrel enhances ventilation in ondine patients: Animal data involving serotoninergic systems. Neuropharmacology. 2016 Aug;107:339-350. doi: 10.1016/j.neuropharm.2016.03.041. Epub 2016 Apr 1. PubMed PMID: 27040794.<br />
6: Regidor PA, Colli E, Schindler AE. Drospirenone as estrogen-free pill and hemostasis: coagulatory study results comparing a novel 4 mg formulation in a 24 + 4 cycle with desogestrel 75 μg per day. Gynecol Endocrinol. 2016 Sep;32(9):749-751. Epub 2016 Mar 30. PubMed PMID: 27028425.<br />
7: Kancheva Landolt N, Bunupuradah T, Kosalaraksa P, Ubolyam S, Thammajaruk N, Cremers S, Zott R, Kerr S, Ananworanich J; HIV-NAT 176 study. High Variability of Hormonal Levels and No Clinically Relevant Interaction Between Ethinyl Estradiol, Desogestrel and Lopinavir/Ritonavir in a Small Sample of HIV-positive Adolescents. J Acquir Immune Defic Syndr. 2016 Aug 15;72(5):507-12. doi: 10.1097/QAI.0000000000000997. PubMed PMID: 26990825.<br />
8: Brache V, Cochon L, Duijkers IJ, Levy DP, Kapp N, Monteil C, Abitbol JL, Klipping C. A prospective, randomized, pharmacodynamic study of quick-starting a desogestrel progestin-only pill following ulipristal acetate for emergency contraception. Hum Reprod. 2015 Dec;30(12):2785-93. doi: 10.1093/humrep/dev241. Epub 2015 Sep 23. PubMed PMID: 26405263.<br />
9: Duijkers IJ, Heger-Mahn D, Drouin D, Skouby S. A randomised study comparing the effect on ovarian activity of a progestogen-only pill (POP) containing desogestrel and a new POP containing drospirenone in a 24/4 regimen. Eur J Contracept Reprod Health Care. 2015;20(6):419-27. doi: 10.3109/13625187.2015.1044082. Epub 2015 Jun 15. PubMed PMID: 26073333.<br />
10: Bitzer J, Banal-Silao MJ, Ahrendt HJ, Restrepo J, Hardtke M, Wissinger-Graefenhahn U, Trummer D. Hormone withdrawal-associated symptoms with ethinylestradiol 20 μg/drospirenone 3 mg (24/4 regimen) versus ethinylestradiol 20 μg/desogestrel 150 μg (21/7 regimen). Int J Womens Health. 2015 May 18;7:501-9. doi: 10.2147/IJWH.S77942. eCollection 2015. PubMed PMID: 26056491; PubMed Central PMCID: PMC4445871.
|