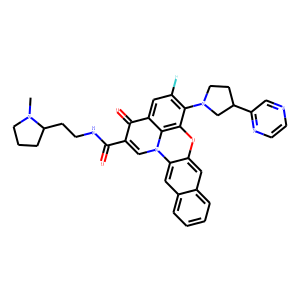
| InChI | InChI=1S/C35H33FN6O3/c1-40-13-4-7-24(40)8-10-39-35(44)26-20-42-29-15-21-5-2-3-6-22(21)16-30(29)45-34-31(42)25(33(26)43)17-27(36)32(34)41-14-9-23(19-41)28-18-37-11-12-38-28/h2-3,5-6,11-12,15-18,20,23-24H,4,7-10,13-14,19H2,1H3,(H,39,44)/t23?,24-/m0/s1 |
| Reference | 1. Cancer Res. 2009 Oct 1;69(19):7653-61. doi: 10.1158/0008-5472.CAN-09-1304. Epub
2009 Sep 8.
<br><br>
Anticancer activity of CX-3543: a direct inhibitor of rRNA biogenesis.
<br><br>
Drygin D(1), Siddiqui-Jain A, O/’Brien S, Schwaebe M, Lin A, Bliesath J, Ho CB,
Proffitt C, Trent K, Whitten JP, Lim JK, Von Hoff D, Anderes K, Rice WG.
<br>
Author information: <br>
(1)Cylene Pharmaceuticals, Inc., San Diego, CA 92121, USA.
<br>
Hallmark deregulated signaling in cancer cells drives excessive ribosome
biogenesis within the nucleolus, which elicits unbridled cell growth and
proliferation. The rate-limiting step of ribosome biogenesis is synthesis of rRNA
(building blocks of ribosomes) by RNA Polymerase I (Pol I). Numerous kinase
pathways and products of proto-oncogenes can up-regulate Pol I, whereas tumor
suppressor proteins can inhibit rRNA synthesis. In tumorigenesis, activating
mutations in certain cancer-associated kinases and loss-of-function mutations in
tumor suppressors lead to deregulated signaling that stimulates Pol I
transcription with resultant increases in ribosome biogenesis, protein synthesis,
cell growth, and proliferation. Certain anticancer therapeutics, such as
cisplatin and 5-fluorouracil, reportedly exert, at least partially, their
activity through disruption of ribosome biogenesis, yet many prime targets for
anticancer drugs within the ribosome synthetic machinery of the nucleolus remain
largely unexploited. Herein, we describe CX-3543, a small molecule
nucleolus-targeting agent that selectively disrupts nucleolin/rDNA G-quadruplex
complexes in the nucleolus, thereby inhibiting Pol I transcription and inducing
apoptosis in cancer cells. CX-3543 is the first G-quadruplex interactive agent to
enter human clinical trials, and it is currently under evaluation against
carcinoid/neuroendocrine tumors in a phase II clinical trial.
<br>
|