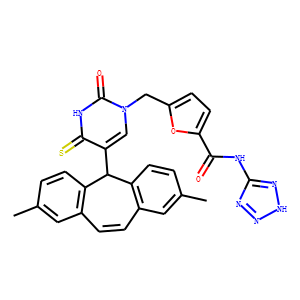
| InChI | InChI=1S/C28H23N7O3S/c1-15-3-8-20-17(11-15)5-6-18-12-16(2)4-9-21(18)24(20)22-14-35(28(37)30-26(22)39)13-19-7-10-23(38-19)25(36)29-27-31-33-34-32-27/h3-12,14,24H,13H2,1-2H3,(H,30,37,39)(H2,29,31,32,33,34,36) |
| Reference | 1. Am J Respir Cell Mol Biol. 2004 Oct;31(4):446-55. Epub 2004 Jul 1.
<br>
Nucleotide-mediated mucin secretion from differentiated human bronchial
epithelial cells.
<br>
Kemp PA(1), Sugar RA, Jackson AD.
<br>
Author information: <br>
(1)Novartis Respiratory Research Centre, Wimblehurst Road, Horsham, West Sussex
RH12 5AB, UK.
<br>
Most current cell-based models for examining the regulation of mucin secretion
demonstrate low signal-to-noise ratios, making experimental manipulation and data
interpretation difficult. Using adenosine triphosphate (ATP) as a mucin
secretagogue, we have developed a model of agonist-induced mucin secretion in
differentiated human bronchial epithelial cells. Mucin secretory signals were
estimated using enzyme-linked lectin assay, and typical signals of 300-400% of
baseline were observed in response to a 30-min exposure to ATP (100 microM). ATP
and uridine triphosphate equipotently stimulated mucin secretion consistent with
mediation via P2Y2 receptor activation. Suramin and AR-C118925XX, a competitive
P2Y2 receptor antagonist, inhibited adenosine 5/’-o-(3-thiotriphosphate)
(ATP-gammaS)-induced mucin secretion. A selective Gq G-protein antagonist
(GP-ANT)-2A completely abrogated ATP-gammaS-induced mucin secretion. Pertussis
toxin and the G(i/o)-specific, GP-ANT-2, had no effect. The phospholipase C
inhibitor, D609, and the protein kinase C inhibitor, calphostin C, substantially
inhibited ATP-gammaS-induced mucin secretion. Phorbol myristate acetate also
stimulated mucin secretion in a calphostin C-sensitive manner. ATP-gammaS-induced
mucin secretion was inhibited by the Ca2+ chelator, 1,2-bis(o-aminophenoxy)
ethane-N,N,N/’,N/’-tetra-acetic acid tetra (acetoxymethyl) ester. Ionomycin and
thapsigargin both stimulated mucin secretion. Our data are broadly consistent
with known G-protein-coupling and downstream signaling events associated with the
P2Y2 receptor. The exceptional signal-to-noise ratios obtained using this model
have permitted clear evaluation of the involvement of these mechanisms in
agonist-induced mucin secretion from differentiated human bronchial epithelial
cells.
|