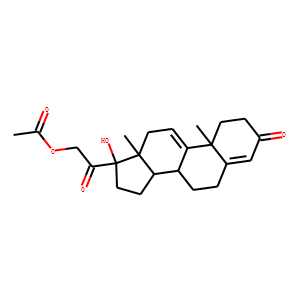
| InChI | InChI=1S/C23H30O5/c1-14(24)28-13-20(26)23(27)11-8-19-17-5-4-15-12-16(25)6-9-21(15,2)18(17)7-10-22(19,23)3/h7,12,17,19,27H,4-6,8-11,13H2,1-3H3/t17-,19+,21+,22+,23+/m1/s1 |
| Reference | </br>1:Identification of PDE6D as a molecular target of anecortave acetate via a methotrexate-anchored yeast three-hybrid screen. Shepard AR, Conrow RE, Pang IH, Jacobson N, Rezwan M, Rutschmann K, Auerbach D, Sriramaratnam R, Cornish VW.ACS Chem Biol. 2013 Mar 15;8(3):549-58. doi: 10.1021/cb300296m. Epub 2013 Jan 9. PMID: 23301619 </br>2:Treatment of steroid-induced elevated intraocular pressure with anecortave acetate: a randomized clinical trial. Stalmans I, Callanan DG, Dirks MS, Moster MR, Robin AL, Van Calster J, Scheib SA, Dickerson JE Jr, Landry TA, Bergamini MV.J Ocul Pharmacol Ther. 2012 Dec;28(6):559-65. doi: 10.1089/jop.2012.0063. Epub 2012 Aug 3. PMID: 22860637 Free PMC Article</br>3:In vitro transport and partitioning of AL-4940, active metabolite of angiostatic agent anecortave acetate, in ocular tissues of the posterior segment. Missel P, Chastain J, Mitra A, Kompella U, Kansara V, Duvvuri S, Amrite A, Cheruvu N.J Ocul Pharmacol Ther. 2010 Apr;26(2):137-46. doi: 10.1089/jop.2009.0132. PMID: 20415622 Free PMC Article</br>4:Suppression and reduction of corticosteroid-induced ocular hypertension by anecortave in sheep. Kaufman PL.Arch Ophthalmol. 2010 Mar;128(3):365-6. doi: 10.1001/archophthalmol.2009.410. No abstract available. PMID: 20212210 </br>5:Suppression of corticosteroid-induced ocular hypertension in sheep by anecortave. Candia OA, Gerometta R, Millar JC, Podos SM.Arch Ophthalmol. 2010 Mar;128(3):338-43. doi: 10.1001/archophthalmol.2009.387. PMID: 20212205 </br>6:Gelatinase expression in retinoblastoma: modulation of LH(BETA)T(AG) retinal tumor development by anecortave acetate. Bajenaru ML, Piña Y, Murray TG, Cebulla CM, Feuer W, Jockovich ME, Marin Castaño ME.Invest Ophthalmol Vis Sci. 2010 Jun;51(6):2860-4. doi: 10.1167/iovs.09-4500. Epub 2010 Jan 27. PMID: 20107171 Free PMC Article</br>7:Hypotensive effect of juxtascleral administration of anecortave acetate in different types of glaucoma. Prata TS, Tavares IM, Mello PA, Tamura C, Lima VC, Belfort R.J Glaucoma. 2010 Sep;19(7):488-92. doi: 10.1097/IJG.0b013e3181c4b0e8. PMID: 20051887 </br>8:Time-lapse imaging of retinal angiogenesis reveals decreased development and progression of neovascular sprouting by anecortave desacetate. Unoki N, Murakami T, Ogino K, Nukada M, Yoshimura N.Invest Ophthalmol Vis Sci. 2010 May;51(5):2347-55. doi: 10.1167/iovs.09-4158. Epub 2009 Dec 3. PMID: 19959648 </br>9:Reduction of intraocular pressure with anecortave acetate in eyes with ocular steroid injection-related glaucoma. Robin AL, Suan EP, Sjaarda RN, Callanan DG, Defaller J; Alcon Anecortave Acetate for IOP Research Team..Arch Ophthalmol. 2009 Feb;127(2):173-8. doi: 10.1001/archophthalmol.2008.595. PMID: 19204235 </br>10:[Present status of studies on the treatment of choroidal neovascularization by Anecortave acetate]. Li X, Wang YS.Zhonghua Yan Ke Za Zhi. 2008 Jun;44(6):571-4. Review. Chinese. PMID: 19035252
|