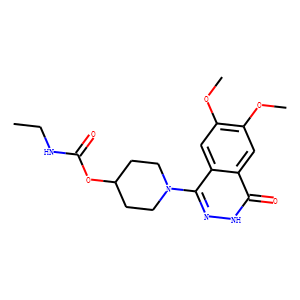
| Reference | 1. Drug Metab Dispos. 2014 Dec;42(12):2049-57. doi: 10.1124/dmd.114.060368. Epub
2014 Sep 23.
<br>
Aldehyde oxidase activity in fresh human skin.
<br>
Manevski N(1), Balavenkatraman KK(1), Bertschi B(1), Swart P(1), Walles M(1),
Camenisch G(1), Schiller H(1), Kretz O(1), Ling B(1), Wettstein R(1), Schaefer
DJ(1), Pognan F(1), Wolf A(1), Litherland K(2).
<br>
Author information:<br>
(1)Drug Metabolism and Pharmacokinetics (N.M., P.S., M.W., G.C., H.S., O.K.,
K.L.) and Pre-clinical Safety (K.K.B., B.B., F.P., A.W.), Novartis Institutes
for BioMedical Research, Novartis Pharma, Basel, Switzerland; and Department of
Plastic, Reconstructive, Aesthetic and Hand Surgery, University Hospital Basel,
Basel, Switzerland (B.L., R.W., D.J.S.).<br>
(2)Drug Metabolism and Pharmacokinetics (N.M., P.S., M.W., G.C., H.S., O.K.,
K.L.) and Pre-clinical Safety (K.K.B., B.B., F.P., A.W.), Novartis Institutes
for BioMedical Research, Novartis Pharma, Basel, Switzerland; and Department of
Plastic, Reconstructive, Aesthetic and Hand Surgery, University Hospital Basel,
Basel, Switzerland (B.L., R.W., D.J.S.) [email protected].
<br>
Human aldehyde oxidase (AO) is a molybdoflavoenzyme that commonly oxidizes
azaheterocycles in therapeutic drugs. Although high metabolic clearance by AO
resulted in several drug failures, existing in vitro-in vivo correlations are
often poor and the extrahepatic role of AO practically unknown. This study
investigated enzymatic activity of AO in fresh human skin, the largest organ of
the body, frequently exposed to therapeutic drugs and xenobiotics. Fresh,
full-thickness human skin was obtained from 13 individual donors and assayed
with two specific AO substrates: carbazeran and zoniporide. Human skin explants
from all donors metabolized carbazeran to 4-hydroxycarbazeran and zoniporide to
2-oxo-zoniporide. Average rates of carbazeran and zoniporide hydroxylations were
1.301 and 0.164 pmol⋅mg skin(-1)⋅h(-1), resulting in 13 and 2% substrate
turnover, respectively, after 24 hours of incubation with 10 μM substrate.
Hydroxylation activities for the two substrates were significantly correlated
(r(2) = 0.769), with interindividual variability ranging from 3-fold
(zoniporide) to 6-fold (carbazeran). Inclusion of hydralazine, an irreversible
inhibitor of AO, resulted in concentration-dependent decrease of hydroxylation
activities, exceeding 90% inhibition of carbazeran 4-hydroxylation at 100 μM
inhibitor. Reaction rates were linear up to 4 hours and well described by
Michaelis-Menten enzyme kinetics. Comparison of carbazeran and zoniporide
hydroxylation with rates of triclosan glucuronidation and sulfation and
p-toluidine N-acetylation showed that cutaneous AO activity is comparable to
tested phase II metabolic reactions, indicating a significant role of AO in
cutaneous drug metabolism. To our best knowledge, this is the first report of AO
enzymatic activity in human skin.<br>
|