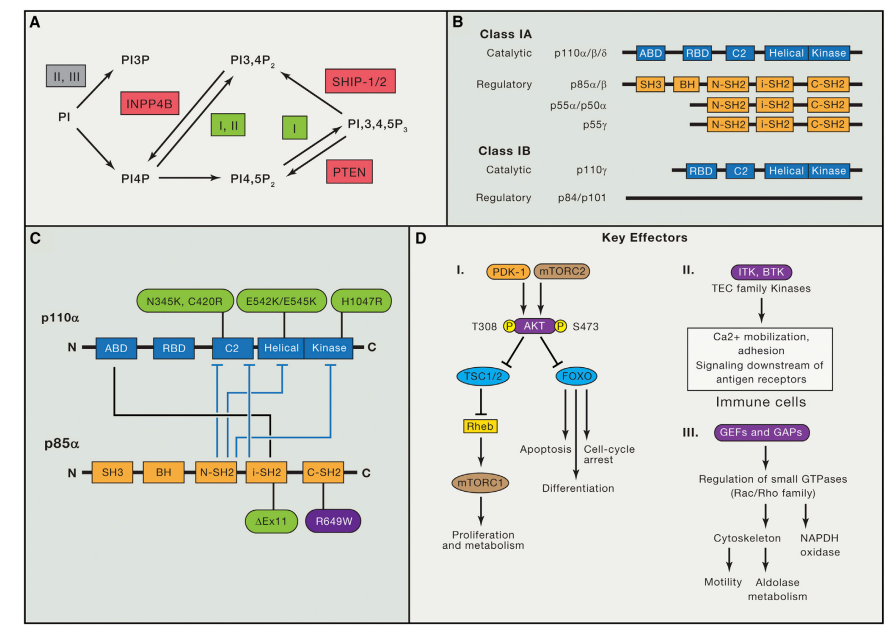
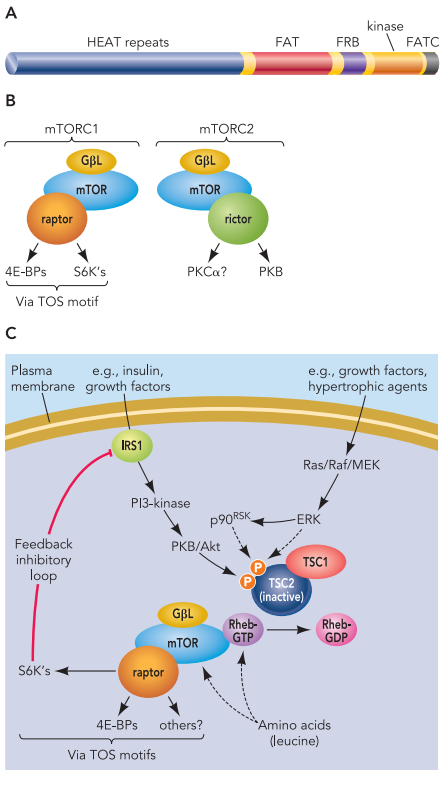
What is the Future of Small Molecule Drugs?
Abstract
In the realm of small-molecule drugs, the future is marked by innovation. Cutting-edge technologies like AI-driven drug design expedite discovery, while personalized therapies tailor treatments to individual genetic profiles. Targeted delivery systems further reduce adverse effects. These advancements offer safer, more potent interventions, reshaping healthcare through heightened precision and improved patient experiences.
As technology continues to advance, a plethora of groundbreaking therapies have surfaced in recent times, presenting novel avenues of treatment to individuals worldwide. However, concurrently, the so-called “conventional” small molecule drugs, often perceived as traditional, are undergoing a continuous process of innovation, rejuvenating their significance. In conjunction with publicly available information, our team aims to acquaint readers with the contemporary significance of small-molecule drugs in the industry. We will also elucidate their inherent developmental strengths and offer insights into their future potential.
Fig.1 Small molecule drug
Small molecule drugs are increasingly innovative
The landscape of small molecule drugs has evolved significantly from those sanctioned decades ago. Propelled by advancements in chemistry and pharmaceutical science, researchers can now craft small-molecule drugs that align more precisely with contemporary medical requisites. For instance, a stark comparison emerges between lenacapavir (marketed as Sunlenca), endorsed just last year for combating multidrug-resistant human immunodeficiency virus (HIV) infections, and the inaugural anti-AIDS drug AZT (zidovudine), sanctioned in 1987. AZT exhibits a considerably simpler chemical configuration, encompassing merely 32 atoms and attainable through a concise sequence of 3-4 chemical reactions. In stark contrast, the elaborately structured lenacapavir, housing 96 atoms, mandates no fewer than 20 intricate synthetic steps for its creation.
The intricate chemical nature of lenacapavir is equally evident in the extensive duration of its development. Spanning from the project’s inception to its FDA approval, Gilead Sciences committed no less than approximately 16 years to this endeavor. Nevertheless, this substantial investment has yielded the fruition of more convenient and efficacious medications for individuals affected by AIDS, thereby notably enhancing their quality of life. Unlike the initial-generation drug AZT, which carries a multitude of adverse reactions, encompassing potential hematological toxicity, myopathy, and lactic acidosis, and necessitates frequent daily administration, lenacapavir presents a stark contrast. Requiring merely biannual dosing, it boasts heightened tolerability and demonstrates efficacy across all identified strains of HIV, including historically recalcitrant variants resistant to other drugs.
Lenacapavir is not an isolated instance of a small molecule drug that confers benefits upon contemporary patients. From the widely prescribed analgesic ibuprofen to the emerging anti-migraine agent atonement, the established cancer chemotherapy medication cyclophosphamide to the advent of the targeted therapy venetoclax, or from statins to the cholesterol-lowering innovation MK-0616 (potentially effective when statins are inadequate), these exemplify the array of small molecule remedies that continue to advance. Amgen’s discovery of the KRAS G12C inhibitor Lumakras (sotorasib, formerly AMG 510) through comprehensive compound library screening serves as a notable breakthrough within the realm of “undruggable” targets. This small molecule drug gained FDA accelerated approval for marketing in 2021. In comparison with bygone pharmaceuticals, today’s small molecule drugs form a novel and enhanced category characterized by larger molecular weights. Although their production and discovery pose increased challenges, they exhibit superior pharmacological attributes that can significantly augment patient care and quality of life.
Advantages of Small Molecule Drugs
Small molecule drugs possess a multitude of therapeutic benefits. For instance, the majority of these drugs exhibit the capability to permeate cell membranes, effectively targeting intracellular proteins. Additionally, their oral bioavailability facilitates direct and efficient drug delivery to patients. In contrast to biological products, the pharmacokinetic and pharmacodynamic profiles of small molecule drugs are more foreseeable, resulting in simplified treatment regimens. This enhanced predictability, along with the stability and oral attributes inherent to small-molecule drugs, empowers patients to conveniently administer their medications virtually anywhere and at any time. This obviates the need for specific storage conditions and significantly bolsters patient adherence to prescribed treatments.
Furthermore, small-molecule drugs exhibit streamlined production and regulatory procedures, facilitating cost-effective mass production and development. This inherent efficiency contributes to the broader dissemination of such drugs, making them instrumental in addressing pressing global medical crises. In specific medical domains, like central nervous system (CNS) disorders, where the presence of the blood-brain barrier poses challenges for alternative treatment modalities, small molecule drugs often emerge as the primary therapeutic option. Notably, over the past five to six years, small-molecule drugs have maintained a prominent presence, constituting 70% of the FDA’s new drug approvals. Presently, within the biomedical sector, small-molecule drugs continue to dominate, accounting for nearly 90% of the overall drug landscape.
Future Outlook for Small Molecule Drugs
With the advancement of artificial intelligence (AI) and machine learning (ML) technologies, coupled with the analysis of extensive databases, substantial strides are anticipated shortly within the domain of target identification, drug combinations, patient screening, and efficacy prediction for small molecule drugs. This progress seeks to refine the development of more precise and well-tolerated medications, all while mitigating the emergence of drug resistance. For instance, in August of the preceding year, Professor David Baker’s team from the University of Washington published a groundbreaking article in the journal Cell. Utilizing an AI technology platform, they achieved the meticulous design of a macrocyclic polypeptide molecule, entirely capable of penetrating cell membranes, thereby introducing a novel avenue for crafting oral medications. This innovative approach is exemplified by the startup company Vilya, co-founded by members of Professor Baker’s team. Notably, Vilya secured a substantial Series A financing of $50 million, led by the esteemed venture capital firm ARCH Venture Partners. This infusion of funds will facilitate the utilization of their cutting-edge technology to develop a new generation of oral therapies meticulously tailored to target specific disease biology. By circumventing traditional high-throughput screening methods, this approach enables the direct synthesis of potential drug candidates, expediting the process.
In addition to macrocyclic polypeptide molecules, the new generation of small molecule drugs also includes “heterobifunctional molecule” drugs. The two ends of this type of molecule can recognize different targets, which in turn can pull together two proteins that would otherwise have no intersection. For example, one end of PROTAC molecules binds to the target protein, and the other end recognizes the E3 ligase in the ubiquitin degradation system. Through this mechanism, the latter can tag target proteins with ubiquitin, facilitating their degradation. This drug development concept of pulling two different proteins together is called induced proximity. The bispecific molecules developed based on this concept can not only pull the E3 ubiquitin ligase near the target protein, but also Pulling deubiquitinase, phosphorylase, and phosphatase to the vicinity of the target protein, thereby stabilizing the target protein and changing the function of the target protein, is expected to overcome the challenges of many undruggable targets. Professor Craig M. Crews, a pioneer in the field of protein degradation, co-founded Arvinas and Halda Therapeutics in 2013 and this year, respectively, to accelerate the scientific transformation of this type of technology, among which Arvinas developed targeting androgen receptors and estrogen receptors The protein degradation agent has been verified by clinical trials.
Distinct treatment modalities inherently come with their own sets of strengths and weaknesses. Hence, the collaborative focus of the industry should be on the formulation of drugs or therapeutic combinations that can optimally cater to the diverse needs of patients across various indications and demographics. Gazing ahead, the realm of small molecule drugs maintains its unique and unparalleled advantages in the sphere of medical treatment. As science and technology perpetually evolve and achieve breakthroughs, it is with eager anticipation that we await the emergence of a broader array of drugs, meticulously developed and optimized, enabling a greater multitude of patients to reap their benefits.