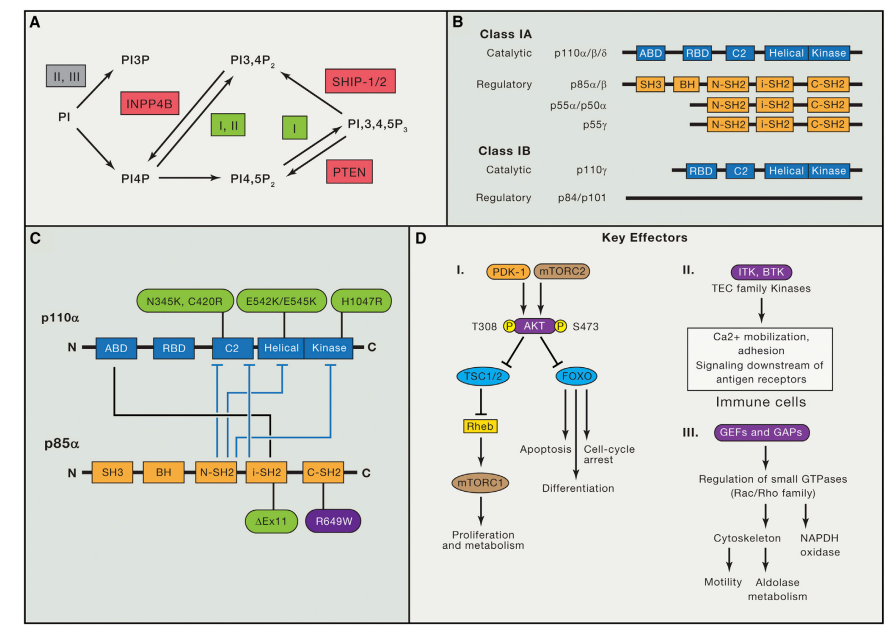
Revolutionizing NSCLC Treatment: Latest Advances in Targeted Therapy
Abstract
The advancements in targeted therapy for non-small cell lung cancer (NSCLC) have significantly transformed treatment approaches, offering more effective and personalized options. However, several challenges hinder the overall success of these therapies. Drug resistance remains a major obstacle, as tumors develop mechanisms to evade treatment. Tumor heterogeneity further complicates treatment, requiring personalized medicine approaches to tailor therapies to individual genetic profiles. Combining targeted therapies with other modalities, such as chemotherapy or immunotherapy, shows promise but also introduces complexities like increased toxicity. Additionally, the high cost of targeted therapies limits access for many patients, raising concerns about equitable healthcare. Future directions include the development of next-generation inhibitors, advances in liquid biopsy techniques, and the integration of artificial intelligence to enhance personalized treatment regimens. Addressing these challenges is crucial for improving patient outcomes and the effectiveness of NSCLC targeted therapies.
Keywords: Non-small cell lung cancer (NSCLC); Targeted therapy; Drug resistance; Tumor heterogeneity; Combination therapies
Introduction
Non-small cell lung cancer (NSCLC) is the most prevalent type of lung cancer, accounting for approximately 85% of all lung cancer cases. NSCLC itself is an umbrella term that includes several subtypes, primarily adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. The primary challenge with NSCLC is its late-stage diagnosis; most patients are diagnosed at an advanced stage where the prognosis is poor. According to recent statistics, the five-year overall survival rate for patients with advanced NSCLC is around 17.4% .
Traditional treatment methods for NSCLC include surgery, chemotherapy, and radiation therapy. Surgery is typically considered for early-stage NSCLC, where it can potentially cure the disease. However, only about 15% of patients are diagnosed at an early stage, making surgery a less viable option for the majority. Chemotherapy and radiation therapy have been the mainstay treatments for advanced stages but come with significant side effects and often only provide modest improvements in survival rates.
The emergence of targeted therapy has brought a significant shift in NSCLC treatment paradigms. Targeted therapies work by specifically attacking cancer cells’ genetic mutations and proteins that promote cancer growth and survival, sparing normal cells and thereby reducing side effects. This approach has shown promising results, particularly in patients with specific genetic alterations such as EGFR mutations and ALK rearrangements .
In conclusion, while traditional treatments for NSCLC have limitations, the development of targeted therapies offers a new avenue for improving patient outcomes. As research continues to advance, these therapies may provide more effective and less toxic options for managing NSCLC.
Traditional Treatment Methods for NSCLC
Traditional treatment methods for non-small cell lung cancer (NSCLC) have primarily included surgery, chemotherapy, and radiation therapy. These conventional approaches have been the cornerstone of lung cancer management for decades, yet each has its specific applications and limitations.
Surgery is often the treatment of choice for early-stage NSCLC, where the tumor is localized and can be removed. Surgical options include lobectomy, pneumonectomy, and segmentectomy, depending on the tumor’s size and location. Surgery aims to remove the entire tumor, offering a potential cure. However, only about 15% of NSCLC cases are diagnosed at an early stage, making surgery less feasible for the majority of patients who present with advanced disease.
Chemotherapy involves using drugs to kill rapidly dividing cancer cells and is typically employed in more advanced stages of NSCLC or as an adjuvant therapy post-surgery to eliminate residual cancer cells. Common chemotherapy regimens include combinations of platinum-based drugs such as cisplatin or carboplatin with other agents like paclitaxel or pemetrexed. Despite its effectiveness in reducing tumor size and prolonging survival, chemotherapy is associated with significant side effects, including nausea, fatigue, and increased infection risk due to its impact on normal dividing cells.
Radiation therapy uses high-energy radiation to destroy cancer cells and shrink tumors. It can be applied as a primary treatment, particularly in patients who are not surgical candidates, or as a palliative measure to relieve symptoms in advanced stages. While radiation therapy can be effective in controlling local tumor growth, it also carries risks of side effects such as radiation pneumonitis and esophagitis.
In conclusion, while traditional treatments like surgery, chemotherapy, and radiation therapy have been fundamental in managing NSCLC, they come with various challenges and limitations, particularly in the advanced stages of the disease. The advent of newer therapies, including targeted therapies and immunotherapy, offers hope for more effective and less toxic treatment options for NSCLC patients.
Breakthroughs in Targeted Therapy for NSCLC
The advent of targeted therapy has revolutionized the treatment landscape for non-small cell lung cancer (NSCLC). Unlike traditional therapies, targeted therapies specifically attack cancer cells based on their genetic mutations and protein expressions, minimizing damage to healthy cells and reducing side effects. This approach has shown significant promise, particularly in patients with specific genetic alterations. Here are some key breakthroughs in targeted therapies for NSCLC:
1. VEGF Inhibitors
Vascular endothelial growth factor (VEGF) inhibitors, such as Bevacizumab and Ramucirumab, have been pivotal in NSCLC treatment. VEGF plays a crucial role in angiogenesis, the formation of new blood vessels that tumors need to grow. Bevacizumab, an anti-VEGF antibody, has been shown to improve survival when combined with chemotherapy. Ramucirumab, another anti-VEGF antibody, also demonstrated efficacy in combination with chemotherapy, providing another valuable option for patients with advanced NSCLC.
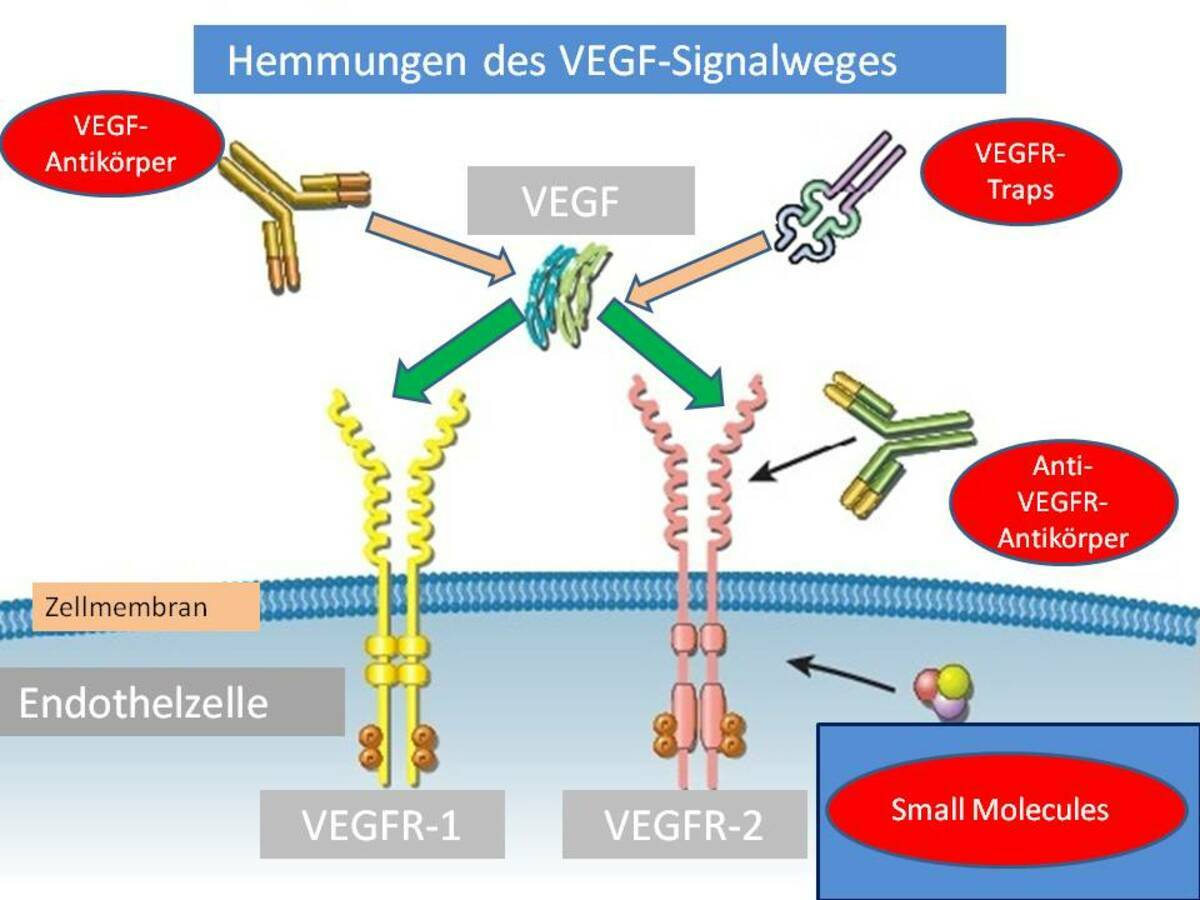
Fig.1 VEGF Inhibitors
2. KRAS Inhibitors
KRAS mutations, found in about 25% of NSCLC cases, have long been considered undruggable. However, recent advancements have led to the development of KRAS inhibitors like Sotorasib and Adagrasib. Sotorasib was the first FDA-approved drug targeting KRAS G12C mutations and has shown promising results in clinical trials. Adagrasib is another KRAS G12C inhibitor with favorable outcomes in early-phase trials, marking a new era in targeting this challenging mutation.
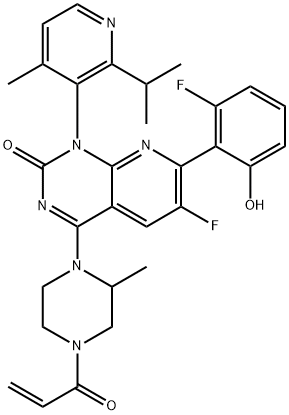
Fig. 2 Structure of Sotorasib
3. EGFR Inhibitors
Mutations in the epidermal growth factor receptor (EGFR) are common in NSCLC, particularly in adenocarcinomas. Drugs like Gefitinib, Erlotinib, and Afatinib target these mutations and have significantly improved outcomes for patients with EGFR-positive NSCLC. These tyrosine kinase inhibitors (TKIs) block the signaling pathways that promote cancer cell proliferation and survival, leading to increased progression-free survival and overall survival.
4. ALK Inhibitors
Anaplastic lymphoma kinase (ALK) rearrangements are present in a subset of NSCLC patients. Crizotinib was the first ALK inhibitor approved and showed substantial efficacy in ALK-positive NSCLC. Subsequent generations of ALK inhibitors, such as Ceritinib and Alectinib, have further improved patient outcomes by overcoming resistance to earlier treatments and demonstrating better brain penetration.
Fig. 3 Structure of Crizotinib
5. ROS1 Inhibitors
ROS1 rearrangements, similar to ALK, are found in a small percentage of NSCLC cases. Crizotinib, also effective against ROS1, was the first-line treatment for ROS1-positive NSCLC. Newer drugs like Lorlatinib have shown efficacy in overcoming resistance to Crizotinib and treating brain metastases, offering hope for better management of ROS1-positive NSCLC.
6. BRAF Inhibitors
BRAF mutations, particularly the V600E mutation, occur in about 2-4% of NSCLC cases. Vemurafenib and Dabrafenib, initially developed for melanoma, have shown effectiveness in NSCLC. Combining BRAF inhibitors with MEK inhibitors like Trametinib has further enhanced treatment efficacy, providing a robust strategy against BRAF-mutant NSCLC.
7. RET Inhibitors
RET fusions are rare but significant in NSCLC. Selective RET inhibitors such as Pralsetinib and Selpercatinib have shown remarkable activity in RET-positive NSCLC. These drugs specifically target RET alterations, offering new therapeutic options for patients with these mutations.
8. MET Inhibitors
MET amplifications and exon 14 skipping mutations are implicated in NSCLC. Capmatinib and Tepotinib, MET inhibitors, have demonstrated significant antitumor activity in MET-altered NSCLC, providing new avenues for targeted treatment.
9. NTRK Inhibitors
NTRK gene fusions, although rare in NSCLC, can be targeted by drugs like Larotrectinib and Entrectinib. These inhibitors have shown high response rates and durable responses, making them valuable options for patients with NTRK fusion-positive NSCLC.
10. PD-1/PD-L1 Inhibitors
Immunotherapy targeting the PD-1/PD-L1 pathway has transformed NSCLC treatment. Nivolumab and Pembrolizumab are PD-1 inhibitors that enhance the body’s immune response against cancer cells. These drugs have shown significant improvement in survival for patients with advanced NSCLC and have become a cornerstone in the treatment landscape.
Challenges and Future Directions in Targeted Therapy for NSCLC
Despite significant advancements in targeted therapy for non-small cell lung cancer (NSCLC), several challenges remain that hinder the overall effectiveness of these treatments. Addressing these issues is crucial for improving patient outcomes and developing more robust therapeutic strategies.
1. Drug Resistance
One of the most significant challenges in targeted therapy is the development of drug resistance. Tumors often develop resistance mechanisms, such as secondary mutations in the target gene, activation of alternative signaling pathways, or phenotypic changes like epithelial-mesenchymal transition (EMT). For example, while EGFR inhibitors like gefitinib and erlotinib initially show high efficacy, many patients eventually develop resistance due to secondary mutations like T790M. Understanding and overcoming these resistance mechanisms is critical for the long-term success of targeted therapies.
2. Tumor Heterogeneity
Tumor heterogeneity, both intertumoral and intratumoral, poses another challenge. NSCLC tumors can exhibit a wide range of genetic mutations and molecular profiles, even within the same patient. This heterogeneity can lead to differential responses to therapy, making it difficult to achieve uniform treatment success. Personalized medicine approaches, including comprehensive genomic profiling, are necessary to tailor treatments to the specific genetic makeup of each tumor.
3. Combination Therapies
Combining targeted therapies with other treatment modalities, such as chemotherapy, immunotherapy, or other targeted agents, holds promise for overcoming resistance and improving efficacy. For instance, combining EGFR inhibitors with MET or VEGF inhibitors has shown potential in preclinical and clinical studies. However, combination therapies also come with increased risk of toxicity and complex drug interactions, necessitating careful management and patient monitoring.
4. Access and Cost
The high cost of targeted therapies can limit access for many patients. These therapies are often expensive, and not all patients have insurance coverage or financial means to afford them. Ensuring equitable access to these life-saving treatments is a significant concern that needs to be addressed through healthcare policy and support programs.
Future Directions
Research is ongoing to develop next-generation inhibitors that can overcome resistance mechanisms and target a broader range of mutations. Advances in liquid biopsy techniques for real-time monitoring of tumor evolution and resistance development are also promising. Additionally, integrating artificial intelligence and machine learning to predict treatment responses and personalize therapy regimens could revolutionize NSCLC treatment in the future.
References
American Cancer Society. (2022). Cancer Facts & Figures 2022.
National Cancer Institute. (2021). SEER Cancer Statistics Review, 1975-2018.