| Reference | [1]. Pharmacol Res. 2015 Jan;91:15-28. doi: 10.1016/j.phrs.2014.10.009. Epub 2014 Nov 10.<br />
Tranilast: a review of its therapeutic applications.<br />
Darakhshan S(1), Pour AB(2).<br />
Author information: (1)Department of Biology, Faculty of Science, Razi University, Kermanshah, Iran. (2)Department of Biology, Faculty of Science, Razi University, Kermanshah, Iran. Electronic address: [email protected].<br />
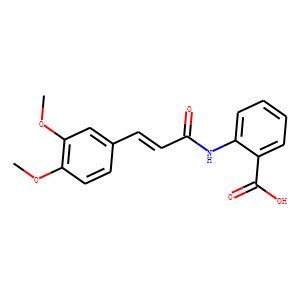
Tranilast (N-[3',4'-dimethoxycinnamoyl]-anthranilic acid) is an analog of a tryptophan metabolite. Initially, tranilast was identified as an anti-allergic agent, and used in the treatment of inflammatory diseases, such as bronchial asthma, atypical dermatitis, allergic conjunctivitis, keloids and hypertrophic scars. Subsequently, the results showed that it could be also effective in the management of a wide range of conditions. The beneficial effects of tranilast have also been seen in a variety of disease states, such as fibrosis, proliferative disorders, cancer, cardiovascular problems, autoimmune disorders, ocular diseases, diabetes and renal diseases. Moreover, several trials have shown that it has very low adverse effects and it is generally well tolerated by patients. In this review, we have attempted to accurately summarize previously published studies relating to the use of tranilast for a range of disorders and discuss the drug's possible mode of action. The major mode of the drug's efficacy appears to be the suppression of the expression and/or action of the TGF-β pathway, but the drug affects other factors as well. The findings presented in this review demonstrate the potential of tranilast for the control of a vast array of pathological situations, furthermore, it is a prescribed drug without severe side effects.<br />
DOI: 10.1016/j.phrs.2014.10.009 PMID: 25447595<br />
<br />
[2]. EMBO Mol Med. 2018 Apr;10(4):e8689. doi: 10.15252/emmm.201708689.<br />
Tranilast directly targets NLRP3 to treat inflammasome-driven diseases.<br />
Huang Y(1)(2), Jiang H(1), Chen Y(3), Wang X(1), Yang Y(4), Tao J(5), Deng X(3), Liang G(6), Zhang H(1)(2), Jiang W(7), Zhou R(7)(2).<br />
Author information: (1)Institute of Immunology and the CAS Key Laboratory of Innate Immunity and Chronic Disease, CAS Center for Excellence in Molecular Cell Sciences, Hefei National Laboratory for Physical Sciences at Microscale, University of Science and Technology of China, Hefei, China. (2)Innovation Center for Cell Signaling Network, University of Science and Technology of China, Hefei, China. (3)State Key Laboratory of Cellular Stress Biology, Innovation Center for Cell Signaling Network, School of Life Sciences, Xiamen University, Xiamen, Fujian, China. (4)Department of Clinical Laboratory, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China. (5)Department of Rheumatology & Immunology, The First Affiliated Hospital of University of Science and Technology of China, Hefei, Anhui, China. (6)CAS Key Laboratory of Soft Matter Chemistry, Department of Chemistry, University of Science and Technology of China, Hefei, China. (7)Institute of Immunology and the CAS Key Laboratory of Innate Immunity and Chronic Disease, CAS Center for Excellence in Molecular Cell Sciences, Hefei National Laboratory for Physical Sciences at Microscale, University of Science and Technology of China, Hefei, China [email protected] [email protected].<br />
The dysregulation of NLRP3 inflammasome can cause uncontrolled inflammation and drive the development of a wide variety of human diseases, but the medications targeting NLRP3 inflammasome are not available in clinic. Here, we show that tranilast (TR), an old anti-allergic clinical drug, is a direct NLRP3 inhibitor. TR inhibits NLRP3 inflammasome activation in macrophages, but has no effects on AIM2 or NLRC4 inflammasome activation. Mechanismly, TR directly binds to the NACHT domain of NLRP3 and suppresses the assembly of NLRP3 inflammasome by blocking NLRP3 oligomerization. In vivo experiments show that TR has remarkable preventive or therapeutic effects on the mouse models of NLRP3 inflammasome-related human diseases, including gouty arthritis, cryopyrin-associated autoinflammatory syndromes, and type 2 diabetes. Furthermore, TR is active ex vivo for synovial fluid mononuclear cells from patients with gout. Thus, our study identifies the old drug TR as a direct NLRP3 inhibitor and provides a potentially practical pharmacological approach for treating NLRP3-driven diseases.<br />
DOI: 10.15252/emmm.201708689 PMCID: PMC5887903 PMID: 29531021<br />
<br />
[3]. Oncol Rep. 2019 Jul;42(1):176-188. doi: 10.3892/or.2019.7150. Epub 2019 May 6.<br />
Tranilast enhances the effect of anticancer agents in osteosarcoma.<br />
Nakashima T(1), Nagano S(1), Setoguchi T(2), Sasaki H(1), Saitoh Y(1), Maeda S(2), Komiya S(1), Taniguchi N(1).<br />
Author information: (1)Department of Orthopaedic Surgery, Graduate School of Medical and Dental Sciences, Kagoshima University, Kagoshima 890‑8520, Japan. (2)Department of Medical Joint Materials, Graduate School of Medical and Dental Sciences, Kagoshima University, Kagoshima 890‑8520, Japan.<br />
Tranilast [N‑(3',4'‑dimethoxycinnamoyl)‑anthranilic acid], initially developed as an antiallergic drug, also exhibits a growth inhibitory effect on various types of cancer. Osteosarcoma is treated mainly with high‑dose methotrexate, doxorubicin, cisplatin and ifosfamide; however, 20‑30% of patients cannot be cured of metastatic disease. We investigated whether tranilast enhances the anticancer effects of chemotherapeutic drugs and analyzed its mechanism of action in osteosarcomas. Tranilast inhibited proliferation of HOS, 143B, U2OS and MG‑63 osteosarcoma cells in a dose‑dependent manner, as well as enhancing the effects of cisplatin and doxorubicin. The average combination index at effect levels for tranilast in combination with cisplatin was 0.57 in HOS, 0.4 in 143B, 0.39 in U2OS and 0.51 in MG‑63 cells. Tranilast and cisplatin synergistically inhibited the viability of osteosarcoma cells. In flow cytometric analysis, although tranilast alone did not induce significant apoptosis, the combination of tranilast and cisplatin induced early and late apoptotic cell death. Expression of cleaved caspase‑3, cleaved poly(ADP‑ribose) polymerase and p‑H2AX was enhanced by tranilast in combination with cisplatin. Tranilast alone increased expression of p21 and Bim protein in a dose‑dependent manner. Cell cycle analysis using flow cytometry demonstrated that the combination of tranilast and cisplatin increased the number of cells in the G2/M phase. Compared with cisplatin alone, the combination increased levels of phospho‑cyclin‑dependent kinase 1 (Y15). In the 143B xenograft model, tumor growth was significantly inhibited by combined tranilast and cisplatin compared with the controls, whereas cisplatin alone did not significantly inhibit tumor growth. In conclusion, tranilast has a cytostatic effect on osteosarcoma cells and enhances the effect of anticancer drugs, especially cisplatin. Enhanced sensitivity to cisplatin was mediated by increased apoptosis through G2/M arrest. Since tranilast has been clinically approved and has few adverse effects, clinical trials of osteosarcoma chemotherapy in combination with tranilast are expected.<br />
DOI: 10.3892/or.2019.7150 PMCID: PMC6549073 PMID: 31059083<br />
<br />
[4]. Eur J Pharmacol. 2018 Jan 5;818:235-240. doi: 10.1016/j.ejphar.2017.10.057. Epub 2017 Oct 28.<br />
Tranilast inhibits interleukin-33 production by macrophages.<br />
Hiraide S(1), Yanagawa Y(2), Iizuka K(1).<br />
Author information: (1)Department of Pharmacology, School of Pharmaceutical Sciences, Health Sciences University of Hokkaido, Kanazawa 1757, Ishikari-Tobetsu 061-0293, Japan. (2)Department of Pharmacology, School of Pharmaceutical Sciences, Health Sciences University of Hokkaido, Kanazawa 1757, Ishikari-Tobetsu 061-0293, Japan. Electronic address: [email protected].<br />
Tranilast is an anti-allergy medication that inhibits the release of chemical mediators such as histamine. However, the mechanisms underlying its anti-allergy effects are not fully understood. Interleukin (IL)-33, a novel member of the IL-1 cytokine family, promotes T helper type 2 immune responses and plays a pathogenic role in allergic disorders. In the present study, we examined the effects of tranilast on IL-33 production by RAW264.7 macrophages. Lipopolysaccharide (LPS) increased both IL-33 mRNA expression and IL-33 protein synthesis. Tranilast significantly inhibited LPS-induced IL-33 protein production by RAW264.7 macrophages in a dose-dependent manner; these same effects were observed on IL-33 mRNA levels in RAW264.7 macrophages and a primary culture of macrophages. LPS markedly activated Akt in RAW264.7 macrophages, whereas tranilast suppressed LPS-induced Akt activation. The effects of tranilast on Akt activation appeared to be responsible for the decrease in IL-33 production. Our present findings suggest that the inhibition of IL-33 production by tranilast might contribute to the anti-allergy effects of this medication.<br />
DOI: 10.1016/j.ejphar.2017.10.057 PMID: 29107673<br />
<br />
[5]. Pharmacol Res Perspect. 2017 Feb 6;5(2):e00291. doi: 10.1002/prp2.291. eCollection 2017 Apr.<br />
Uricosuric targets of tranilast.<br />
Mandal AK(1), Mercado A(2), Foster A(1), Zandi-Nejad K(3), Mount DB(1).<br />
Author information: (1)Renal Divisions VA Boston Healthcare System and Brigham and Women's Hospital Boston Massachusetts. (2)Renal Divisions Departamento de Nefrología Instituto Nacional de Cardiología Ignacio Chávez Mexico City Mexico. (3)Renal Division Beth Israel Deaconess Medical Center Boston Massachusetts.<br />
Uric acid, generated from the metabolism of purines, has both proven and emerging roles in human disease. Serum uric acid in humans is determined by production and by the net balance of reabsorption and secretion in kidney and intestine. In the human kidney, epithelial reabsorption dominates over secretion, such that in normal subjects there is at least 90% net reabsorption of filtered urate resulting in a fractional excretion of <10%. Tranilast, an anti-inflammatory drug with pleiotropic effects, has a marked hypouricemic, uricosuric effect in humans. We report here that tranilast is a potent inhibitor of [14C]-urate transport mediated by the major reabsorptive urate transporters (URAT1, GLUT9, OAT4, and OAT10) in Xenopus oocytes; this provides an unequivocal molecular mechanism for the drug's uricosuric effect. Tranilast was found to inhibit urate transport mediated by URAT1 and GLUT9 in a fully reversible and noncompetitive (mixed) manner. In addition, tranilast inhibits the secretory urate transporters NPT1, OAT1, and OAT3 without affecting the secretory efflux pump ABCG2. Notably, while benzbromarone and probenecid inhibited urate as well as nicotinate transport, tranilast inhibited the urate transport function of URAT1, GLUT9, OAT4, OAT10, and NPT1, without significantly affecting nicotinate transport mediated by SMCT1 (IC 50 ~1.1 mmol/L), SMCT2 (IC 50 ~1.0 mmol/L), and URAT1 (IC 50 ~178 μmol/L). In summary, tranilast causes uricosuria by inhibiting all the major reabsorptive urate transporters, selectively affecting urate over nicotinate transport. These data have implications for the treatment of hyperuricemia and gout, the pharmacology of tranilast, and the structure-function analysis of urate transport.<br />
DOI: 10.1002/prp2.291 PMCID: PMC5368959 PMID: 28357121
|